Vaccines developed by Pfizer/BioNTech and Moderna are the marquee developments of RNA technology that can potentially immunization programs targeting HIV, malaria, influenza and other viral pathogens.
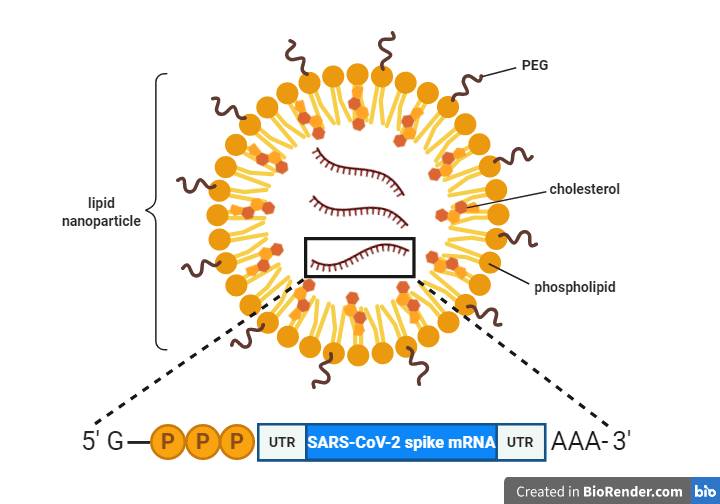
RNA vaccines employ a messenger RNA (mRNA) that encodes the full-length SARS-CoV-2 spike protein. Upon injection into a vaccine recipient, the mRNA enters cells and is translated by the host protein synthesis machinery into the SARS-CoV-2 spike protein.
The vaccines teach the body to recognize and destroy disease-causing agents. In contrast to traditional vaccines (weakened pathogens or antigens – fragments of the proteins or sugars on their surfaces), RNA vaccines carry only the directions for producing these invaders’ proteins. Not the pathogen or fragments.
RNA vaccines produce the antigens, essentially turning the body into its own inoculation factory.
Results of the first successful translations of foreign mRNA in mammals was published in 1990, and this technology has been refined ever since.
RNA stability has been a concern but shown to be remedied through enhanced delivery methods.
Sanofi Pasteur first used RNA to encode an influenza antigen in mice. The RNA produced an immunogenic response, but the lipid delivery system that the team employed was too toxic to use in people. A decade would pass before companies discovered the LNP technologies that would make today’s COVID-19 vaccines possible (see Geall et al. (2012)).
In 2012, the US Defense Advanced Research Projects Agency (DARPA) began funding groups at Novartis, Pfizer, AstraZeneca, Sanofi Pasteur and elsewhere to work on RNA-encoded vaccines and therapeutics. CureVac and Moderna built on that initial work.
References
- Geall, A. J. et al. Proc. Natl Acad. Sci. USA 109, 14604–14609 (2012).