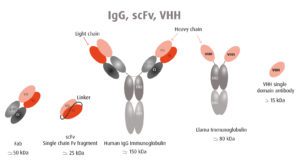
In a posting on Virology (https://www.virology.ws/2021/01/14/camelids-for-covid/), Columbia University Professor Vincent Racaniello makes the cases for mass production of camelid monoclonal antibodies to rapidly suppress CoVid than possible through monoclonal antibodies.
Racaniello argues the antigen binding domain (VHH in the figure) can be produced on its own in a nanobody. Two VHH domains can be linked together to target separate epitopes (scFv in the figure). Since VHH single domain antibodies lack the Fc domain, and therefore cannot bind Fc receptors, this strategy avoids antibody-dependent enhancement.
To produce nanobodies against SARS-CoV-2, an alpaca and a llama were immunized with purified spike protein. Three nanobodies from the alpaca and one from the llama were identified that neutralize virus infection of cells in culture. The results of binding and structural studies revealed that three of the nanobodies recognize an epitope on the spike protein that is distinct from the site recognized by the other nanobody.
Three of the four nanobodies appear to block virus infection of cells by causing the spike protein to change to the post-fusion conformation, which is irreversible. The spike post-fusion conformation is usually attained upon binding to the cell receptor, ACE2, but the nanobodies can trigger fusion in the absence of this protein.
It appears nanobodies can be delivered to the respiratory mucosa by inhalation of aerosols. This delivery method could reduce the dose needed and allow treatment outside of medical facilities.